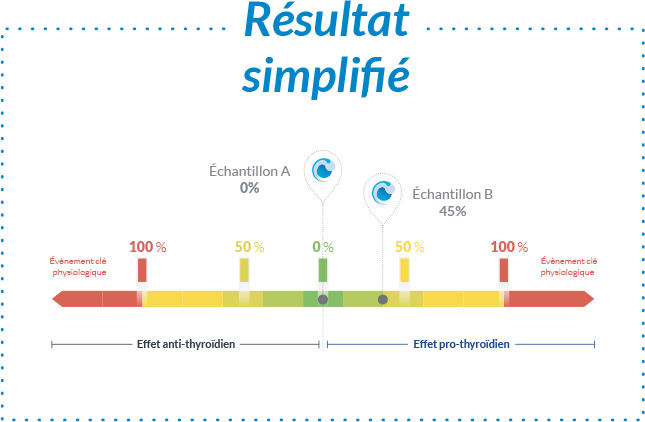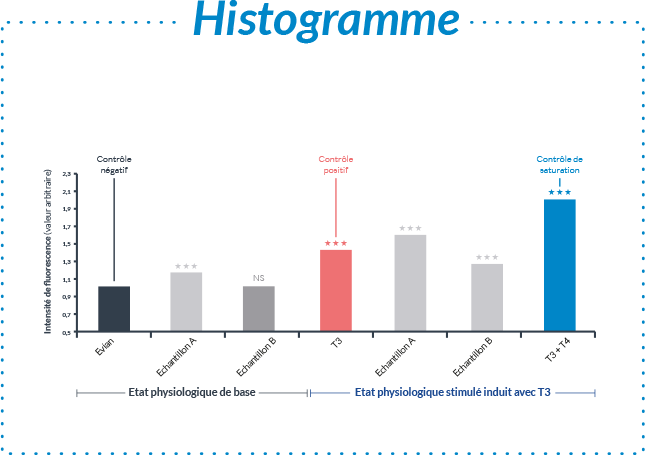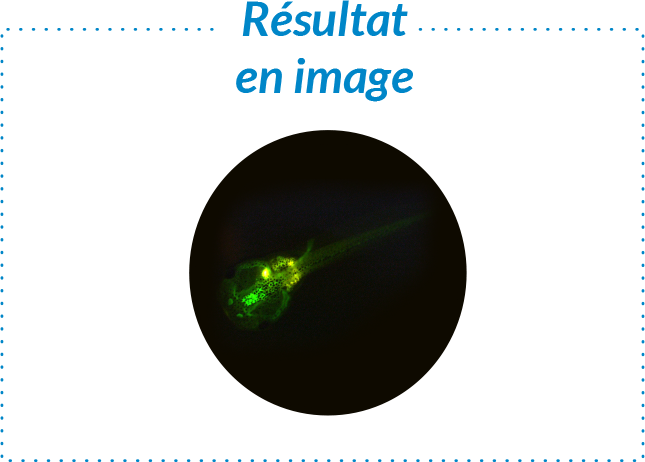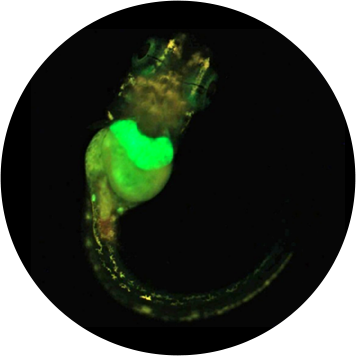
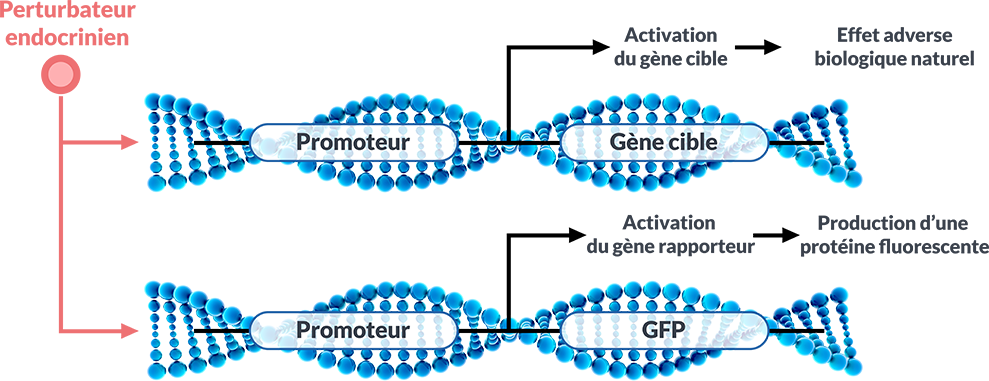
 Pourquoi choisir un test sur organisme entier réalisé in vitro ?
Pourquoi choisir un test sur organisme entier réalisé in vitro ? Quel est l'intérêt de cette technologie ?
Quel est l'intérêt de cette technologie ? Quels axes endocriniens tester (œstrogénique, androgénique ou thyroïdien)?
Quels axes endocriniens tester (œstrogénique, androgénique ou thyroïdien)? Est-ce qu’un test suffit à identifier un perturbateur endocrinien ?
Est-ce qu’un test suffit à identifier un perturbateur endocrinien ? Comment puis-je communiquer sur mes produits ou supports de vente à l'issue d'un test endocrinien ?
Comment puis-je communiquer sur mes produits ou supports de vente à l'issue d'un test endocrinien ?